Articles
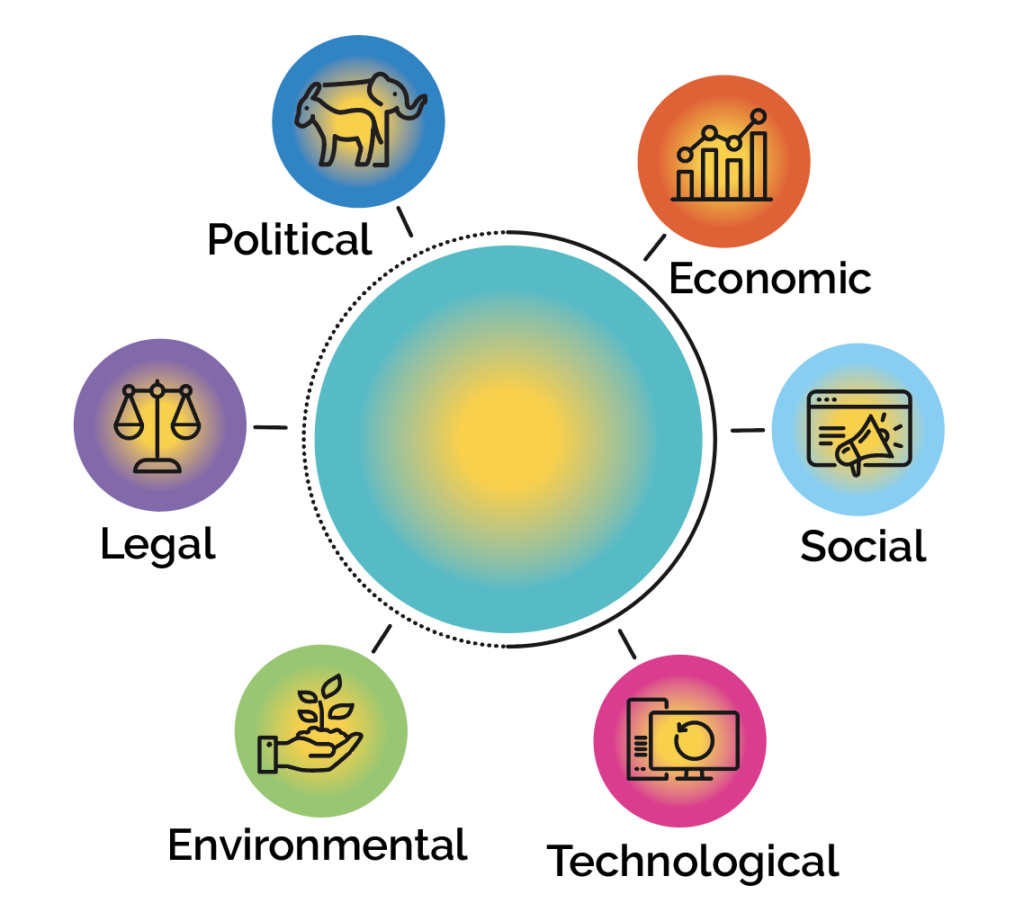
Global Pharmaceutical Oral Solids and Non-Sterile Liquids Market: PESTEL analysis
BACK TO RESOURCE CENTER Other Content Guide to Aseptic ManufacturingGuide to Oral Dose ManufacturingArticlesBrochuresBy The NumbersCase StudiesMeet the ScientistNewslettersPress ReleasesTrade ShowsVideos Global Pharmaceutical Oral Solids and Non-Sterile Liquids Market: PESTEL analysis This analysis highlights the impact that key sectors have on oral solids and non-sterile liquids drug development. Since December 2021, the Biden Administration has…
Read MoreIntegrity Testing and Selecting the Best Closure for Your Lyo Product
BACK TO RESOURCE CENTER Other Content Guide to Aseptic ManufacturingGuide to Oral Dose ManufacturingArticlesBrochuresBy The NumbersCase StudiesMeet the ScientistNewslettersPress ReleasesTrade ShowsVideos Integrity Testing and Selecting the Best Closure for Your Lyo Product By: Bryan Braxton, Ph.D., Senior Director, Aseptic R&D Many approach the container closure systems as part of the formulation. It must be compatible…
Read MoreNorth America, Europe Lead Lyophilization Development
BACK TO RESOURCE CENTER Other Content Guide to Aseptic ManufacturingGuide to Oral Dose ManufacturingArticlesBrochuresBy The NumbersCase StudiesMeet the ScientistNewslettersPress ReleasesTrade ShowsVideos North America, Europe Lead Lyophilization Development North America and Europe are anticipated to be the most lucrative regions in the global lyophilization market owing to the presence of biotechnology and pharmaceutical companies and the…
Read MoreOutsourcing Analytical Testing is a Money Saver
BACK TO RESOURCE CENTER Other Content Guide to Aseptic ManufacturingGuide to Oral Dose ManufacturingArticlesBrochuresBy The NumbersCase StudiesMeet the ScientistNewslettersPress ReleasesTrade ShowsVideos Outsourcing Analytical Testing is a Money Saver By: Rahul Mehta, MSc, Associate Director The life science industry has traditionally been successful in weathering uncertain economic conditions thanks to the highly inelastic demand for drug…
Read MoreParenteral Pestel Analysis
BACK TO RESOURCE CENTER Other Content Guide to Aseptic ManufacturingGuide to Oral Dose ManufacturingArticlesBrochuresBy The NumbersCase StudiesMeet the ScientistNewslettersPress ReleasesTrade ShowsVideos PARENTERAL PESTEL ANALYSIS This analysis highlights the impact that key sectors have on parenteral drug development. Increased spending on biologics, the need for faster route of drug administration, increasing preference for single-dose administration of…
Read MoreLyophilization Pestel Analysis
BACK TO RESOURCE CENTER Other Content Guide to Aseptic ManufacturingGuide to Oral Dose ManufacturingArticlesBrochuresBy The NumbersCase StudiesMeet the ScientistNewslettersPress ReleasesTrade ShowsVideos LYOPHILIZATION PESTEL ANALYSIS This analysis highlights the impact that key sectors have on the lyophilization industry and practices. As lyophilization enhances the shelf life of drugs, in particular vaccines, the freeze drying technique will…
Read MoreThe Cold, Dry Facts of Lyophilization
Partnering with a CDMO like Pii can progress your drug’s parenteral delivery from early-stage development concept to clinic. Pii can fill cartridges for use with a customer’s pen device, for a variety of products, including biologics. Supporting a range of therapies that require frequent injections and variable dosing, the 3mL cartridge in combination with a pen device can improve patient compliance.
Read MoreDeveloping an Effective Lyophilization Cycle
Partnering with a CDMO like Pii can progress your drug’s parenteral delivery from early-stage development concept to clinic. Pii can fill cartridges for use with a customer’s pen device, for a variety of products, including biologics. Supporting a range of therapies that require frequent injections and variable dosing, the 3mL cartridge in combination with a pen device can improve patient compliance.
Read MoreFive Trends Driving Lyophilization
Partnering with a CDMO like Pii can progress your drug’s parenteral delivery from early-stage development concept to clinic. Pii can fill cartridges for use with a customer’s pen device, for a variety of products, including biologics. Supporting a range of therapies that require frequent injections and variable dosing, the 3mL cartridge in combination with a pen device can improve patient compliance.
Read MorePrecise, Targeted, and Controlled Release of Parenteral Cancer Treatments
Partnering with a CDMO like Pii can progress your drug’s parenteral delivery from early-stage development concept to clinic. Pii can fill cartridges for use with a customer’s pen device, for a variety of products, including biologics. Supporting a range of therapies that require frequent injections and variable dosing, the 3mL cartridge in combination with a pen device can improve patient compliance.
Read More