Contact Us
- Solutions
- Resources
- About
- Contact Us
close
Optional callout banner for highlighted news or events
Learn More
The drug development process is among the most complex, costly, and regulated of human pursuits and the statistical chances of success are horribly low. According to Derek Lowe’s blog, “In The Pipeline”, published by Science Translational Medicine, the chances of success for a drug compound entering Phase 1 trial has remained at slightly under 10% for the last two decades. Mr. Lowe challenges us to consider any other major business category that operates with such a high expectation for failure.
According to an MIT study, of those drugs that enter clinical trials, nearly 14% will ultimately gain FDA approval. Vaccines perform significantly better with a 33.4% chance of success, but investigational oncology is much lower at 3.4%. Despite these dismal chances for success, there is some hopeful news. I know what you’re thinking, please get to the good news?
The study further found that clinical trials employing companion diagnostics tended to be more successful than those that did not. As a deeper understanding of patient populations continues to advance, fueled by healthcare data technology, the chances of success are expected to get better. When we couple this trend in patient disease knowledge with a disciplined phase-appropriate process for drug development, a more cost effective, success-oriented model emerges. Let’s consider phase-appropriate development by applying it to process validation.

A common requirement for all clinical trial drug products for human testing is the need to validate analytical methods, equipment, and utilities as well as other unique drug-specific factors such as environmental monitoring of aseptic processing areas used in sterile filling.
A phase appropriate method to drug process development applies an understanding of “what” is needed and “when” it is needed for each phase of the drug development process to better support the overall Validation Master Plan. The key to achieving a cost effective, success-oriented outcome is building the plan around a logical sequence of events and following it with discipline as you take the drug product through the regulatory process.
For example, Phase 1 often stirs debate regarding the level of testing required to show that clinical batches meet the minimum requirements of USP and the limited specifications established at this early stage. A phase appropriate method will lean toward the minimum requirements. However, there are some process development professionals that will conduct analysis required in later phases: testing blends for uniformity or going beyond USP requirements for tablet or capsule dissolution and uniformity of dosage units.
To be clear, going beyond what FDA requires by phase does not violate establishing a validation track record, but it does expend time and money. Conducting activities in Phase 1 that are aligned with Phase 2 and beyond consumes resources that may not be necessary if Phase 1 is not successful. Using resources earmarked for Phase 2 or 3 should be triggered when accomplishing the outcomes of the previous phase, thus delivering cost effectiveness based on success. See Figure 1 for a critical validation activities flowchart by phase.
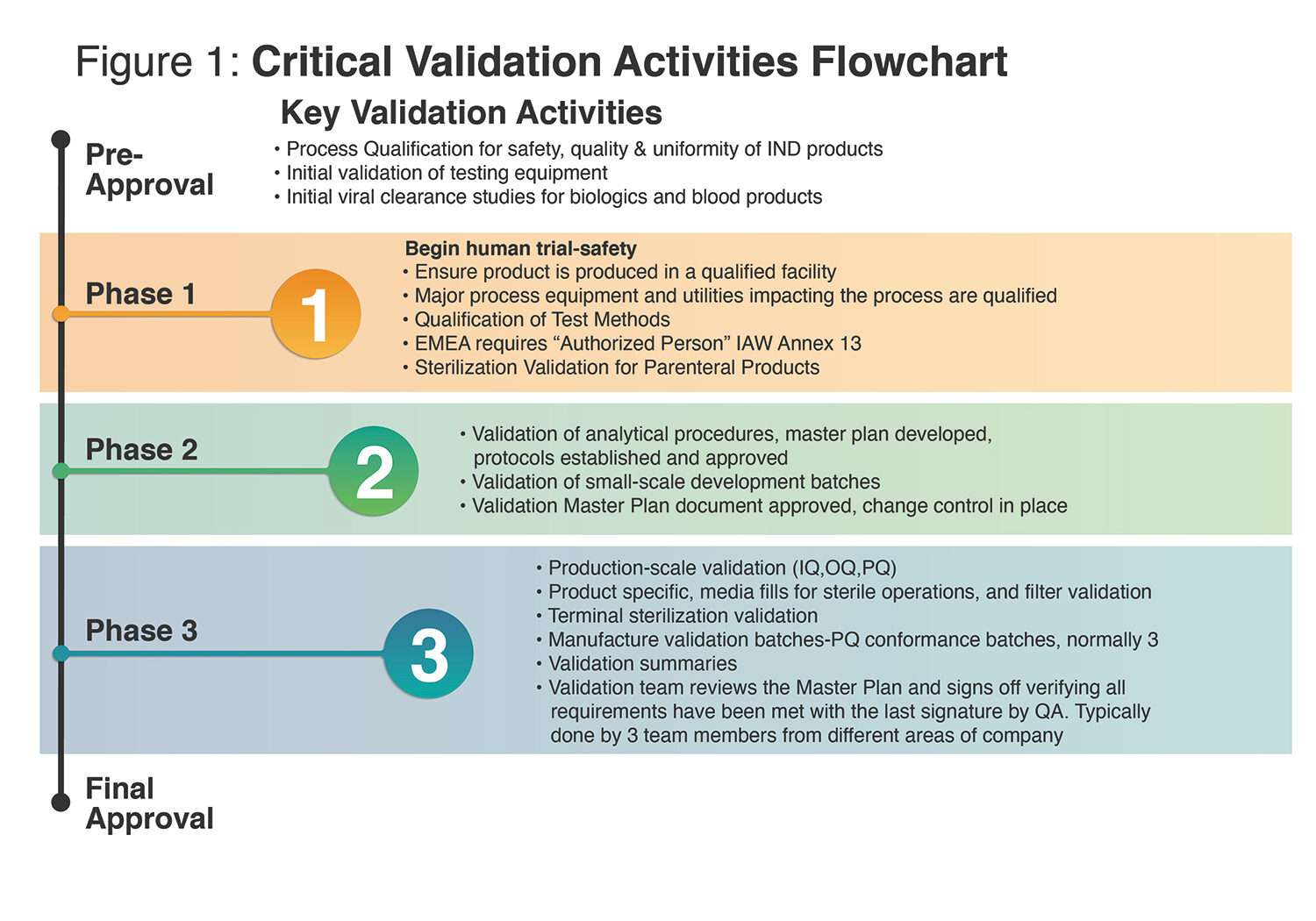
Currently, and sadly, 90% of drug development projects seeking success in Phase 1 clinical trials will fail. With continued gains in patient understanding, companion diagnostics, and healthcare data technology, that low chance of success will become better. However, for now, drug developers can use a phase-appropriate method for process development and FDA regulatory approval to wisely use their finite resources in a way that is prompted by phase-aligned success.
Pharmaceutics International, Inc. (Pii) is a US-based contract development and manufacturing organization (CDMO) located in Hunt Valley, Maryland. The experienced scientists, engineers, and staff at Pii pride themselves on adroitly employing a phase appropriate method of drug development for the prudent use of their customer’s resources as they solve challenging problems. In addition to offering end-to-end development services, Pii manufactures a variety of dosage forms to include complex parenteral drugs and has a wealth of analytical testing capabilities. Its Hunt Valley campus has four aseptic suites with lyophilization capabilities. Our talented professionals stand ready to help!
Like what you read? Share with your network: